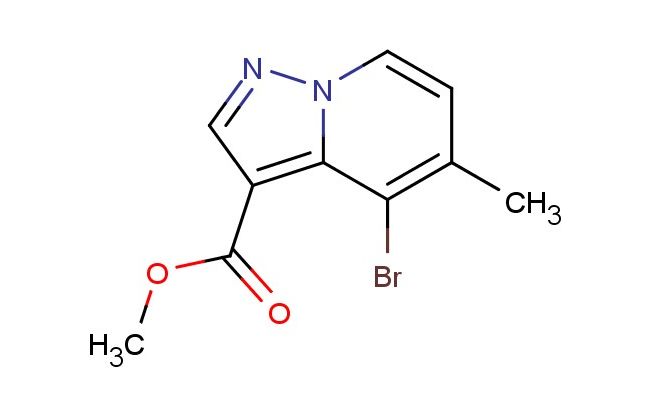
methyl 4-bromo-5-methylpyrazolo[1,5-a]pyridine-3-carboxylate
$300.00
CAS No.: 1345121-21-2
Catalog No.: 192893
Purity: 95%
MF: C10H9BrN2O2
MW: 269.098
Storage: 2-8 degree Celsius
SMILES: BrC=1C=2N(C=CC1C)N=CC2C(=O)OC
Catalog No.: 192893
Purity: 95%
MF: C10H9BrN2O2
MW: 269.098
Storage: 2-8 degree Celsius
SMILES: BrC=1C=2N(C=CC1C)N=CC2C(=O)OC
For R&D use only. Not for human or animal use.
methyl 4-bromo-5-methylpyrazolo[1,5-a]pyridine-3-carboxylate; CAS No.: 1345121-21-2; methyl 4-bromo-5-methylpyrazolo[1,5-a]pyridine-3-carboxylate. PROPERTIES: This compound presents as a white crystalline solid with molecular formula C11H9BrN2O2 and a molecular weight of 287.10 g/mol. It demonstrates a melting point in the range of 118-122 C and shows moderate solubility in common organic solvents like methanol and ethyl acetate. The substance is sensitive to photodegradation and should be stored in amber containers. Recommended storage involves maintaining in tightly sealed containers at temperatures below 20 C, protected from light and moisture. When handling, standard laboratory safety protocols should be followed, including the use of protective gloves and eye wear, as methyl 4-bromo-5-methylpyrazolo[1,5-a]pyridine-3-carboxylate may release toxic fumes upon heating. The bromo group requires careful handling in strongly basic environments. APPLICATIONS: In the pharmaceutical industry, methyl 4-bromo-5-methylpyrazolo[1,5-a]pyridine-3-carboxylate serves as an intermediate for synthesizing anticancer agents, as described in medicinal chemistry literature focusing on kinase inhibitors. Its bromo substituent enables participation in cross-coupling reactions for constructing biaryl systems. Additionally, this compound functions as a building block for preparing agrochemicals with insecticidal activities through nucleophilic aromatic substitution reactions. Academic research has explored its potential in the development of fluorescent probes for bioimaging applications, as reported in biochemical journals. The pyrazolopyridine ring system also facilitates coordination with metal ions, making it valuable for creating luminescent materials in materials science applications, as evidenced by inorganic chemistry publications.
Reviews
Write Your Own Review



![methyl 6-bromo-5-methylpyrazolo[1,5-a]pyridine-3-carboxylate](https://www.chemshuttle.com/media/catalog/product/cache/31dbf0bffbfa69a5826a72cec9a446de/1/9/192892_2.jpg)
![6-bromo-5-methylpyrazolo[1,5-a]pyridine](https://www.chemshuttle.com/media/catalog/product/cache/31dbf0bffbfa69a5826a72cec9a446de/1/9/192894_2.jpg)
![4-bromo-3-methyl-[1,2,3]triazolo[1,5-a]pyridine](https://www.chemshuttle.com/media/catalog/product/cache/eda3e020a548044183bce7a8a96486f8/1/8/185539_6.jpg)
![8-Bromo-5-methyl-[1,2,4]triazolo[4,3-a]pyridine](https://www.chemshuttle.com/media/catalog/product/cache/eda3e020a548044183bce7a8a96486f8/w/l/wlz4378_1.jpg)
![3-{8-bromo-3-oxo-2H,3H-[1,2,4]triazolo[4,3-a]pyridin-2-yl}piperidine-2,6-dione](https://www.chemshuttle.com/media/catalog/product/cache/eda3e020a548044183bce7a8a96486f8/y/q/yqf0126_1.jpg)
![8-bromo-2-methyl-[1,2,4]triazolo[1,5-a]pyridine](https://www.chemshuttle.com/media/catalog/product/cache/eda3e020a548044183bce7a8a96486f8/1/9/199279_4.jpg)
![ethyl 8-bromo-5-oxo-6H-imidazo[1,2-c]pyrimidine-2-carboxylate](https://www.chemshuttle.com/media/catalog/product/cache/eda3e020a548044183bce7a8a96486f8/2/0/201162_2.jpg)
![8-bromo-5-oxo-6H-imidazo[1,2-c]pyrimidine-2-carboxylic acid](https://www.chemshuttle.com/media/catalog/product/cache/eda3e020a548044183bce7a8a96486f8/2/0/201161_2.jpg)
![8-Bromo-5-chloroimidazo[1,2-c]pyrimidine-2-carbonitrile](https://www.chemshuttle.com/media/catalog/product/cache/eda3e020a548044183bce7a8a96486f8/2/0/201160_2.jpg)
![8-bromo-6-chloro-2-methylimidazo[1,2-b]pyridazine](https://www.chemshuttle.com/media/catalog/product/cache/eda3e020a548044183bce7a8a96486f8/1/9/197700_2.jpg)
![8-bromo-6-chloro-2-methyl-[1,2,4]triazolo[1,5-a]pyridine](https://www.chemshuttle.com/media/catalog/product/cache/eda3e020a548044183bce7a8a96486f8/1/9/197680_2.jpg)
![4-bromo-5-methylpyrazolo[1,5-a]pyridine](https://www.chemshuttle.com/media/catalog/product/cache/eda3e020a548044183bce7a8a96486f8/1/9/192895_2.jpg)