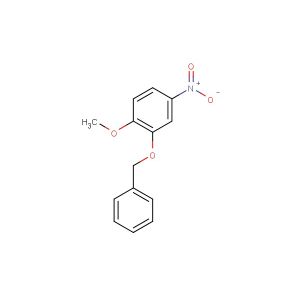
tert-butyl 7-(benzyloxy)-3-(hydroxymethyl)-1H-indole-1-carboxylate
$400.00
CAS No.: 914349-16-9
Catalog No.: 196221
Purity: 95%
MF: C21H23NO4
MW: 353.418
Storage: 2-8 degree Celsius
SMILES: C(C1=CC=CC=C1)OC=1C=CC=C2C(=CN(C12)C(=O)OC(C)(C)C)CO
Catalog No.: 196221
Purity: 95%
MF: C21H23NO4
MW: 353.418
Storage: 2-8 degree Celsius
SMILES: C(C1=CC=CC=C1)OC=1C=CC=C2C(=CN(C12)C(=O)OC(C)(C)C)CO
For R&D use only. Not for human or veterinary use.
tert-butyl 7-(benzyloxy)-3-(hydroxymethyl)-1H-indole-1-carboxylate; CAS No.: 914349-16-9; tert-butyl 7-(benzyloxy)-3-(hydroxymethyl)-1H-indole-1-carboxylate. PROPERTIES: This compound presents a tert-butyl 7-(benzyloxy)-3-(hydroxymethyl)-1H-indole-1-carboxylate structure, combining a tert-butyl ester, a benzyloxy group, a hydroxymethyl substituent, and an indole ring system. It typically appears as a white to off-white crystalline solid with a molecular weight of approximately 363.4 g/mol (C20H21NO4). The melting point ranges between 100-105 C, and it exhibits moderate solubility in common organic solvents like ethyl acetate, dichloromethane, and tetrahydrofuran while being sparingly soluble in water. Proper storage requires a tightly sealed container in a cool, dry place. Safety considerations include wearing appropriate PPE. It is classified as a skin and eye irritant (GHS07) with the hazard statement H315-H319. APPLICATIONS: tert-Butyl 7-(benzyloxy)-3-(hydroxymethyl)-1H-indole-1-carboxylate serves as a specialized intermediate in pharmaceutical research. Its tert-butyl 7-(benzyloxy)-3-(hydroxymethyl)-1H-indole-1-carboxylate structure enables diverse reactivity patterns, including benzylic oxidation, nucleophilic substitution at the hydroxymethyl group, and ester hydrolysis. In medicinal chemistry, it is used to develop bioactive molecules targeting kinases, G protein-coupled receptors, and proteases. The benzyloxy group can be removed by hydrogenolysis to release the parent indole for further functionalization. This compound also functions as a building block in the synthesis of fluorescent probes and bioconjugation reagents. Academic studies utilize it as a model system in Medicinal Chemistry journals, focusing on optimizing the tert-butyl 7-(benzyloxy)-3-(hydroxymethyl)-1H-indole-1-carboxylate scaffold for improved biological activity and pharmacokinetic properties.
Reviews
Write Your Own Review



![3-benzyl-3-azabicyclo[3.1.1]heptan-6-one](https://www.chemshuttle.com/media/catalog/product/cache/eda3e020a548044183bce7a8a96486f8/z/b/zb0417_2.jpg)