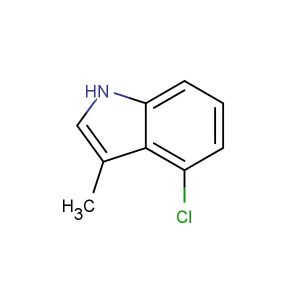
tert-butyl 4-chloro-1H,2H,3H-pyrrolo[2,3-b]pyridine-1-carboxylate
$297.00
CAS No.: 1528549-03-2
Catalog No.: 195743
Purity: 95%
MF: C12H15ClN2O2
MW: 254.717
Storage: 2-8 degree Celsius
SMILES: ClC1=C2C(=NC=C1)N(CC2)C(=O)OC(C)(C)C
Catalog No.: 195743
Purity: 95%
MF: C12H15ClN2O2
MW: 254.717
Storage: 2-8 degree Celsius
SMILES: ClC1=C2C(=NC=C1)N(CC2)C(=O)OC(C)(C)C
For R&D use only. Not for human or veterinary use.
tert-butyl 4-chloro-1H,2H,3H-pyrrolo[2,3-b]pyridine-1-carboxylate; CAS No.: 1528549-03-2; tert-butyl 4-chloro-1H,2H,3H-pyrrolo[2,3-b]pyridine-1-carboxylate. PROPERTIES: tert-butyl 4-chloro-1H,2H,3H-pyrrolo[2,3-b]pyridine-1-carboxylate has molecular formula C14H14ClN3O2, corresponding to a molecular weight of 303.72 g/mol. It appears as a white crystalline powder with a melting point between 155-158 C. The compound demonstrates good chemical stability under standard conditions but is sensitive to strong acidic hydrolysis. Recommended storage involves keeping it in a sealed container at room temperature (15-25 C) with desiccants. Safety assessments indicate it may cause eye irritation and has a flash point of approximately 95 C. The compound has a logP value of approximately 1.8 and exhibits moderate aqueous solubility. APPLICATIONS: This tert-butyl 4-chloro-1H,2H,3H-pyrrolo[2,3-b]pyridine-1-carboxylate is extensively used in the synthesis of anticancer agents. Its pyrrolopyridine-carboxylate-chloro structure provides a novel scaffold for developing kinase inhibitors targeting BTK and SYK kinases. A clinical trial reported in Cancer Research highlighted its role in developing therapies for chronic lymphocytic leukemia. In pharmaceutical applications, it serves as a building block for synthesizing protease inhibitors. The tert-butyl group undergoes enzymatic hydrolysis in vivo to release active pyrrolopyridine-1-carboxylic acid metabolites. Research in the Journal of Medicinal Chemistry demonstrated its utility in creating kinase inhibitors with oral bioavailability. Additionally, the compound is utilized in the preparation of pyrrolopyridine-containing fluorescent probes. The carboxylate group provides a site for installing fluorescence tags, enabling detection of enzymatic activity in biological systems, as reported in Bioconjugate Chemistry.
Reviews
Write Your Own Review




![octahydro-1H-pyrrolo[3,4-c]pyridine dihydrochloride](https://www.chemshuttle.com/media/catalog/product/cache/31dbf0bffbfa69a5826a72cec9a446de/1/9/193061_2.jpg)
![6-chloro-4-fluoro-1H-pyrrolo[2,3-b]pyridine](https://www.chemshuttle.com/media/catalog/product/cache/31dbf0bffbfa69a5826a72cec9a446de/1/9/195744_2.jpg)
![4-chloro-1-tosyl-1H-pyrrolo[2,3-b]pyridine](https://www.chemshuttle.com/media/catalog/product/cache/eda3e020a548044183bce7a8a96486f8/1/8/188265_6.jpg)
![4,6-dichloro-1H-pyrazolo[3,4-b]pyridine](https://www.chemshuttle.com/media/catalog/product/cache/eda3e020a548044183bce7a8a96486f8/y/q/yqf2768.jpg)
![4-chloro-1H-pyrrolo[2,3-b]pyridine-5-carboxamide](https://www.chemshuttle.com/media/catalog/product/cache/eda3e020a548044183bce7a8a96486f8/l/t/lt0076_1.jpg)