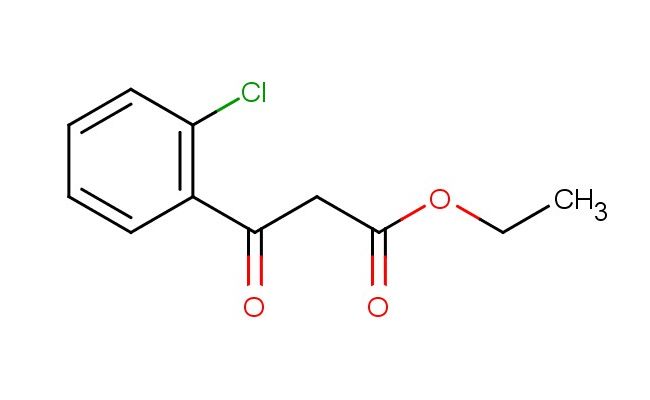
ethyl 3-(2-chlorophenyl)-3-oxopropanoate
$300.00
CAS No.: 19112-35-7
Catalog No.: LT0129
Purity: 95%
MF: C11H11ClO3
MW: 226.659
Storage: 2-8 degree Celsius
SMILES: ClC1=C(C=CC=C1)C(CC(=O)OCC)=O
Catalog No.: LT0129
Purity: 95%
MF: C11H11ClO3
MW: 226.659
Storage: 2-8 degree Celsius
SMILES: ClC1=C(C=CC=C1)C(CC(=O)OCC)=O
For R&D use only. Not for human or veterinary use.
CAS NO.: 19112-35-7;ethyl 3-(2-chlorophenyl)-3-oxopropanoate. PROPERTIES: This chlorinated ester presents as a colorless liquid with a molecular weight of approximately 222.6 g/mol. The ethyl 3-(2-chlorophenyl)-3-oxopropanoate combines an ethyl ester with a chlorophenyl substituent and a ketone functionality. It exhibits limited aqueous solubility but good dissolution in ethyl acetate and methanol. Stability characterization reveals sensitivity to base-catalyzed hydrolysis of the ester group and light exposure, necessitating storage at 2-8 degree Celsius in amber glass containers. Handlers should use powder hoods with HEPA filtration and wear cut-resistant gloves during handling. Skin contact may cause mild irritation requiring thorough washing. Inhalation may induce respiratory tract irritation; treatment includes fresh air and medical evaluation. Eye exposure requires extended rinsing and possible corticosteroid application. Waste should be hydrolyzed with dilute acid prior to disposal. APPLICATIONS: The ethyl 3-(2-chlorophenyl)-3-oxopropanoate serves as a key intermediate in the synthesis of various pharmaceuticals. Its alpha-keto ester framework provides opportunities for nucleophilic addition reactions. Research teams utilize this compound as a starting material for creating beta-lactam antibiotics and nonsteroidal anti-inflammatory agents. The chlorophenyl group enhances metabolic resistance in resulting drug candidates. Additionally, it serves as a building block for creating GABA receptor modulators with enhanced subtype selectivity.
Reviews
Write Your Own Review



![(2',4'-dichloro-[1,1'-biphenyl]-4-yl)methanamine 2,2,2-trifluoroacetic acid](https://www.chemshuttle.com/media/catalog/product/cache/eda3e020a548044183bce7a8a96486f8/2/0/203990_3.jpg)
![(2',4'-dichloro-[1,1'-biphenyl]-4-yl)methanamine](https://www.chemshuttle.com/media/catalog/product/cache/eda3e020a548044183bce7a8a96486f8/1/9/197540_13.jpg)