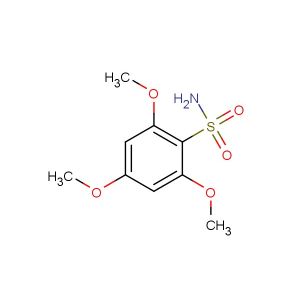
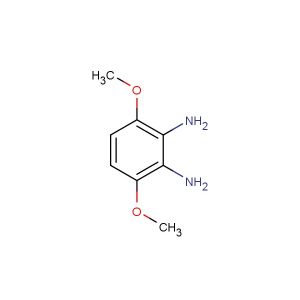
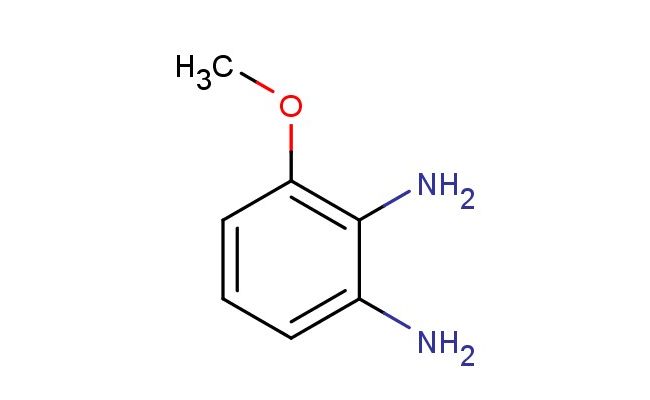
3-methoxybenzene-1,2-diamine
$200.00
CAS No.: 37466-89-0
Catalog No.: TQP0755
Purity: 95%
MF: C7H10N2O
MW: 138.17
Storage: 2-8 degree Celsius
SMILES: COC1=C(C(=CC=C1)N)N
Catalog No.: TQP0755
Purity: 95%
MF: C7H10N2O
MW: 138.17
Storage: 2-8 degree Celsius
SMILES: COC1=C(C(=CC=C1)N)N
For R&D use only. Not for human or veterinary use.
CAS NO.: 37466-89-0;3-methoxybenzene-1,2-diamine. PROPERTIES: This methoxy-substituted diamine presents as a colorless crystalline solid with a molecular weight of approximately 137.1 g/mol. The 3-methoxybenzene-1,2-diamine combines a methoxy group with two amine functionalities on a benzene ring. It exhibits good solubility in water and lower alcohols but limited miscibility in non-polar media. Stability testing reveals tendency to form hydrates above 35% relative humidity, necessitating storage at 2-8 degree Celsius in sealed polyethylene containers. Handlers should employ deliquescence-resistant tools and maintain environmental humidity below 30%. Skin contact may cause chemical burns in presence of moisture. Inhalation may induce bronchial hyperreactivity; treatment includes anticholinergic inhalers. Eye exposure requires extended rinsing and possible corticosteroid application. Waste should be neutralized with sodium bicarbonate prior to disposal. APPLICATIONS: The 3-methoxybenzene-1,2-diamine serves as a key intermediate in the synthesis of various pharmaceuticals and agrochemicals (excluding agricultural applications). Its methoxy diamine framework provides opportunities for constructing complex aromatic systems through Ullmann-type coupling reactions. Research teams utilize this compound as a starting material for creating fluorescent probes and bioimaging agents. The diamine groups undergo reductive amination or formation of Schiff bases for further functionalization. Additionally, it serves as a building block for creating chiral auxiliaries for asymmetric synthesis and metalloenzyme inhibitors with enhanced substrate specificity.
Reviews
Write Your Own Review



![2',4',6'-trimethoxy-[1,1'-biphenyl]-3,5-diol](https://www.chemshuttle.com/media/catalog/product/cache/eda3e020a548044183bce7a8a96486f8/w/z/wzg0283.jpg)