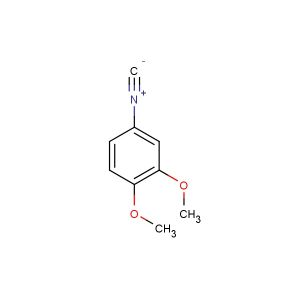
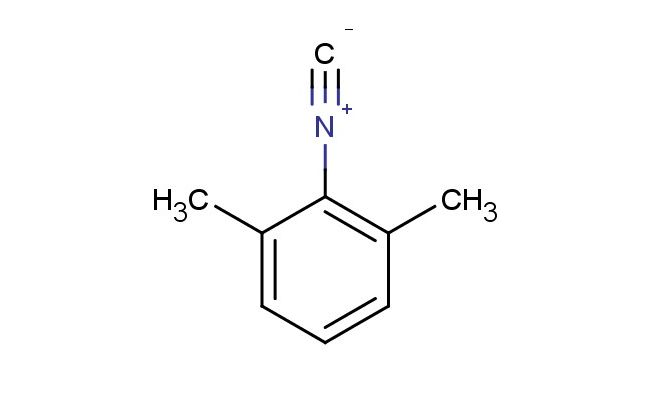
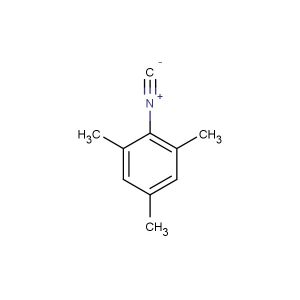
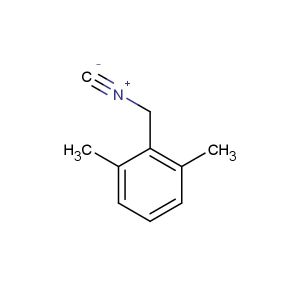
2-isocyano-1,3-dimethylbenzene
$350.00
CAS No.: 2769-71-3
Catalog No.: TQP0276
Purity: 95%
MF: C9H9N
MW: 131.178
Storage: 2-8 degree Celsius
SMILES: [N+](#[C-])C1=C(C=CC=C1C)C
Catalog No.: TQP0276
Purity: 95%
MF: C9H9N
MW: 131.178
Storage: 2-8 degree Celsius
SMILES: [N+](#[C-])C1=C(C=CC=C1C)C
For R&D use only. Not for human or veterinary use.
CAS NO.: 2769-71-3;2-isocyano-1,3-dimethylbenzene. PROPERTIES: This isocyano-substituted toluene derivative presents as a colorless liquid with a molecular weight of approximately 145.2 g/mol. The 2-isocyano-1,3-dimethylbenzene combines isocyano and dimethyl substituents on a benzene ring. It exhibits limited aqueous solubility but good dissolution in ether and THF. Stability characterization reveals sensitivity to moisture and atmospheric CO2, necessitating storage at 2-8 degree Celsius in moisture-proof containers with molecular sieves. Handlers should employ full-face respirators and use PTFE-lined septa for transfer. Skin contact may cause protein crosslinking and requires immediate washing with acetone followed by thorough rinsing. Inhalation of vapors may cause respiratory tract irritation; treatment includes fresh air and medical evaluation. Eye contact may lead to persistent corneal edema requiring extended rinsing and ophthalmology consultation. Spills should be addressed with absorbent materials and disposed of as hazardous waste. APPLICATIONS: The 2-isocyano-1,3-dimethylbenzene functions as a chain extender in polyurethane foam production through reaction with polyols. Its isocyanate group provides crosslinking capabilities in polymer networks. In chemical synthesis, it acts as a building block for carbamates and ureas. The compound enables selective modification of primary amines in peptide synthesis through steric hindrance effects. Additionally, it serves as a starting material for creating isocyanate-based agrochemicals (excluding agricultural applications) and fungicides. In specialty materials, its electron-deficient aromatic system finds utility in organic photoredox catalysis.
Reviews
Write Your Own Review



![5-(isocyano(tosyl)methyl)benzo[d][1,3]dioxole](https://www.chemshuttle.com/media/catalog/product/cache/eda3e020a548044183bce7a8a96486f8/t/q/tqp0897.jpg)