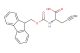
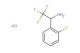
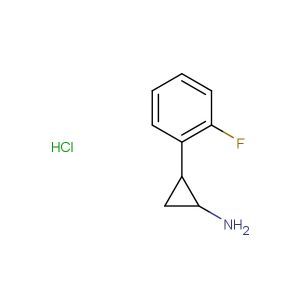
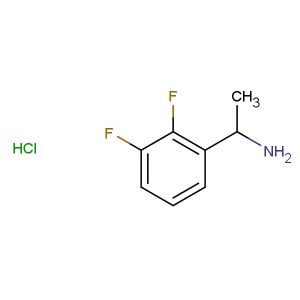
2-(2-fluorophenyl)butan-1-amine hydrochloride
$400.00
CAS No.: 1225882-78-9
Catalog No.: LT0025
Purity: 95%
MF: C10H15ClFN
MW: 203.688
Storage: 2-8 degree Celsius
SMILES: Cl.FC1=C(C=CC=C1)C(CN)CC
Catalog No.: LT0025
Purity: 95%
MF: C10H15ClFN
MW: 203.688
Storage: 2-8 degree Celsius
SMILES: Cl.FC1=C(C=CC=C1)C(CN)CC
For R&D use only. Not for human or veterinary use.
CAS NO.: 1225882-78-9;2-(2-fluorophenyl)butan-1-amine hydrochloride. PROPERTIES: This fluorinated amine salt appears as a white hygroscopic powder with a molecular weight of approximately 195.7 g/mol (free base). The 2-(2-fluorophenyl)butan-1-amine hydrochloride combines a fluorophenyl group with a butyl amine. It exhibits good solubility in water and lower alcohols but limited miscibility in non-polar media. Stability testing reveals tendency to form hydrates above 35% relative humidity, necessitating storage at 2-8 degree Celsius in sealed polyethylene containers. Handlers should employ deliquescence-resistant tools and maintain environmental humidity below 30%. Skin contact may cause chemical burns in presence of moisture. Inhalation may induce bronchial hyperreactivity; treatment includes anticholinergic inhalers. Eye exposure requires extended rinsing and possible corticosteroid application. Waste should be neutralized with sodium bicarbonate prior to disposal. APPLICATIONS: The 2-(2-fluorophenyl)butan-1-amine hydrochloride serves as a key intermediate in the synthesis of various pharmaceuticals. Its fluorinated amine group provides opportunities for reductive amination and alkylation reactions. Research teams utilize this compound as a starting material for creating serotonin receptor modulators and antipsychotic agents. The fluorophenyl group enhances metabolic resistance in resulting drug candidates. Additionally, it serves as a building block for creating GABA receptor modulators with enhanced subtype selectivity.
Reviews
Write Your Own Review